Effective May 27, 2019, empagliflozin (Jardiance™) and empagliflozin with metformin (Synjardy®) are now available for coverage under PharmaCare as limited coverage benefits requiring Special Authority approval. The criteria for coverage is available on the Special Authority website. Jardiance and Synjardy are drugs used in adult patients with Type 2 diabetes and are in a class of medications called Sodium-Glucose Cotransporter 2 (SGLT-2) inhibitors.
Plan W clients who have been grandfathered under the Non-Insured Health Benefits program for SGLT-2 inhibitors (canagliflozin or dapagliflozin with or without metformin) from October 1, 2017 through May 27, 2019 will continue to receive coverage for these medications. Coverage will continue through Plan W.
Plan W Coverage for Insulin Glargine
The PharmaCare announcement on May 27, 2019 affects insulin glargine.
For FNHA clients already receiving the originator insulin glargine (Lantus®), Lantus coverage will continue. Clients are encouraged to switch to its biosimilar version Basaglar™. Since Aug. 2018, all new Plan W clients starting on insulin glargine have been covered for Basaglar. Basaglar remains a regular benefit for FNHA clients meaning no Special Authority is required.
Accessing Coverage
Clients are encouraged to discuss what options are available to them with their healthcare teams.
FNHA is actively supporting the continuity of therapy for SGLT-2 inhibitors and insulin glargine. Clients with any questions or who would like more information are encouraged to contact First Nations Health Benefits at 1-855-550-5454.
The updated FNHA information sheet with the Summary of Diabetes Drugs, Insulins, and Supplies Coverage for FNHA Clients is available on our website:
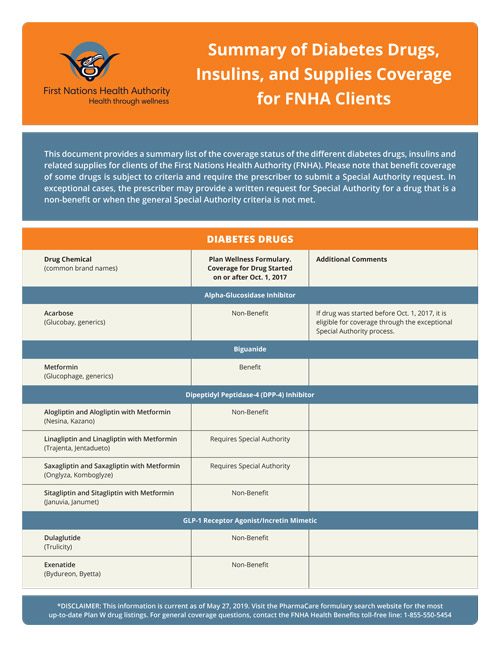
Summary of Diabetes Drugs, Insulins, and Supplies Coverage (PDF 87 KB)
Background Information
PharmaCare announced the expansion coverage on May 27, 2019: https://news.gov.bc.ca/releases/2019HLTH0080-001072